Neurodegenerative dementias are widespread in our aging society and are expected to increase significantly. The most common forms of dementia, such as Alzheimer’s disease and Frontotemporal dementia, are characterized by pathological accumulation of the tau protein in the brain - and are therefore also referred to as tauopathies.
So far, however, there are no therapies that can stop these diseases. One major reason is the lack of informative models for basic research: animal models only inadequately reproduce processes in the human brain and the development of tau pathology; and nerve cell cultures derived from human stem cells only form tau variants typical of young neurons. The tau variants that are decisive for the disease in adult neurons are still missing.
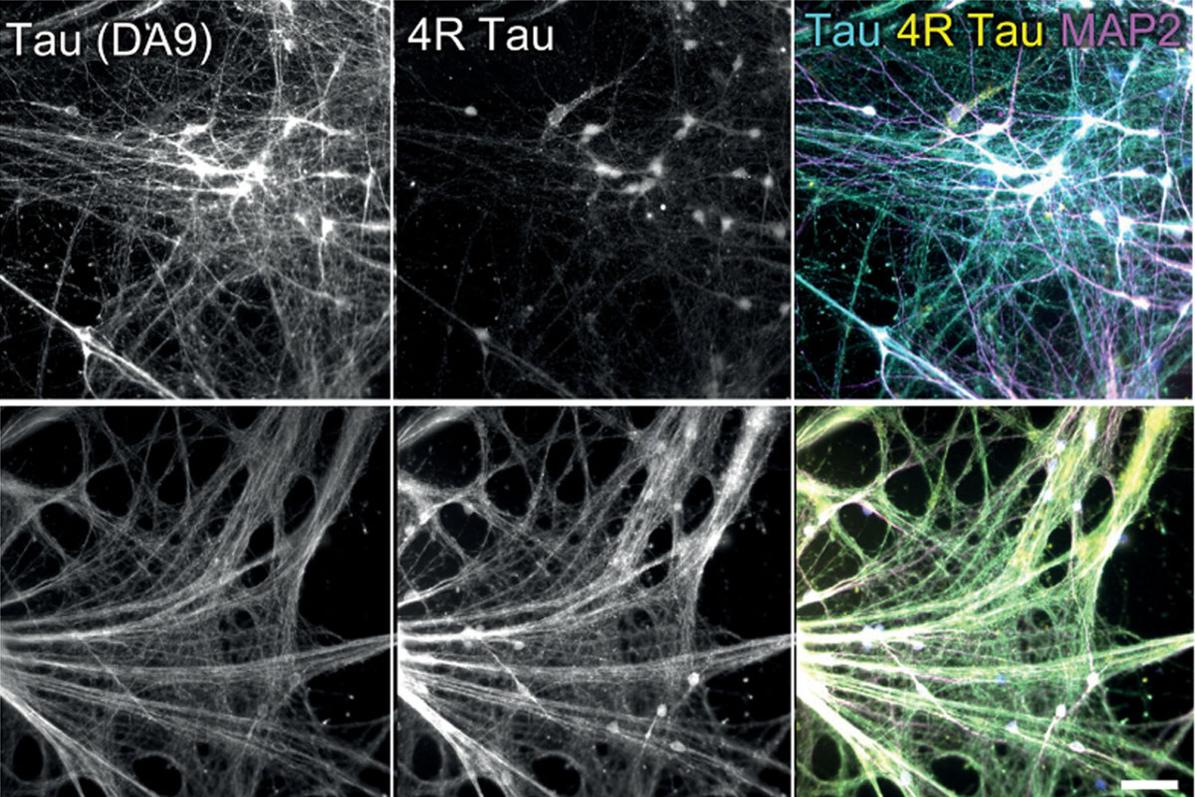
In a research study led by Prof. Dominik Paquet at the Institute for Stroke and Dementia Research at LMU Munich, this problem has now been solved for the first time by developing a human model for tauopathies. Using the gene-editing tool CRISPR/Cas9, first author Dr. Angelika Dannert modified human neurons so that they form the adult tau isoform and carry disease-causing mutations.
Unlike earlier models, these cells independently developed disease features typical of Alzheimer’s disease and other tauopathies, including tau aggregates that closely resemble the deposits found in the brains of affected individuals, known as tangles, as well as signs of neuronal damage such as loss of synapses.
The researchers also showed that their new model is already suitable for drug development. Dr. Dannert states with excitement “...that a compound currently being tested in a clinical trial significantly reduced tau pathology, and that we were also able to successfully test an imaging biomarker for the diagnosis of certain tauopathies!” Prof. Paquet summarized: “...our work closes an important gap between animal experiments and human disease, and the model provides a new platform for developing and testing urgently needed therapies against dementia.”